AI Automation for Mental Health
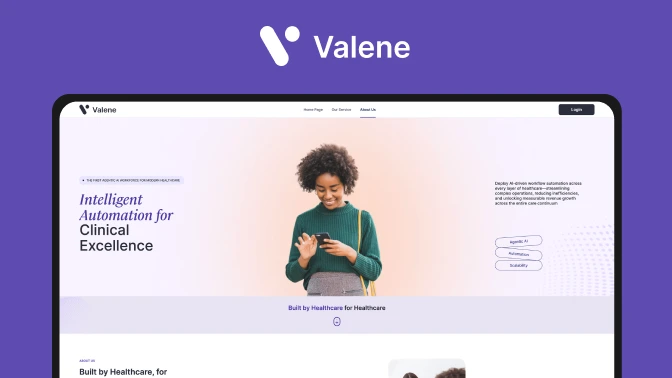
Valene Health is a pioneering telepsychiatry platform that leverages AI to enhance mental healthcare delivery.
A comprehensive lab management software platform that centralizes sample tracking, instrument management, workflow automation, inventory control, quality assurance, and regulatory compliance purpose-built for clinical laboratories, research institutions, pharmaceutical labs, and diagnostic centers that demand accuracy, traceability, and efficiency at every stage of laboratory operations.
Get a Free Consultation
Operational Execellence
Tech Expertise
Consistent Commitement
Based on 43 Reviews
Enable automated workflows, compliance management, performance tracking, and multi-site coordination across research and diagnostic laboratory environments.
Our delivery framework ensures secure, scalable, and compliance-ready execution aligned with laboratory standards and enterprise performance objectives.
Assess laboratory workflows, sample management needs, and compliance requirements to define development scope.
Design scalable system architecture aligned with laboratory operations and data management processes.
Develop laboratory management modules and integrate with lab instruments and healthcare systems.
Ensure the platform meets laboratory standards, data security protocols, and operational reliability.
Deploy the system while enabling continuous monitoring, user training, and operational optimization.
Deploy a lab management software solution engineered for the traceability, compliance, and operational precision your laboratory cannot afford to compromise on.
Talk to Our Experts
Valene Health is a pioneering telepsychiatry platform that leverages AI to enhance mental healthcare delivery.

Advinow is an AI-driven healthcare platform that automates patient engagement and consultation processes, helping healthcare providers deliver efficient, on-demand services while improving operations for urgent care.
View More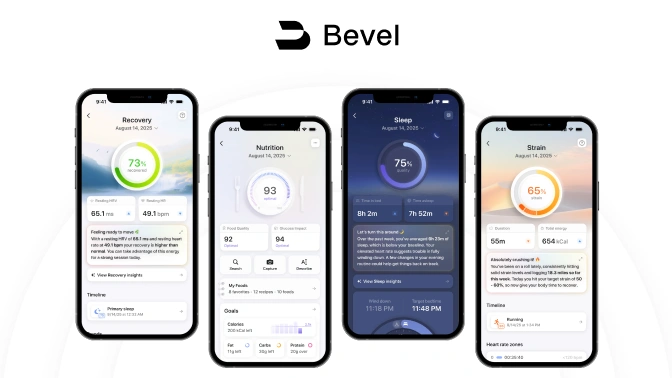
Bevel is a health intelligence platform that unifies wearable and lifestyle data to generate personalized metabolic and wellness insights.
Our Lab management software is fully customizable, supporting diverse laboratory workflows, compliance standards, multi-location operations, and integration requirements.
Yes, the platform supports secure API-based integration with LIS, EHR, billing systems, and laboratory instruments.
Enterprise implementations usually range from 12–24 weeks depending on workflow complexity, integrations, and compliance validation requirements.
Built-in audit trails, role-based access controls, encrypted storage, and automated documentation workflows ensure compliance readiness.
Organizations typically achieve reduced sample errors, faster turnaround times, improved reporting accuracy, and enhanced operational efficiency.