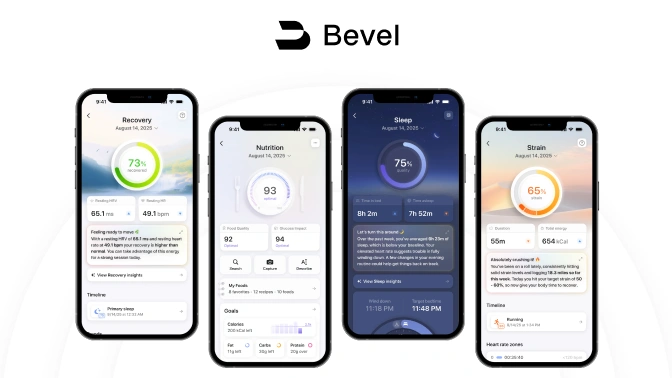
AI Health and Wellness Companion
Bevel is a health intelligence platform that unifies wearable and lifestyle data to generate personalized metabolic and wellness insights.
More InfoA specialized clinical trial management software development company designing and engineering end-to-end CTMS platforms built to streamline trial planning, site management, patient recruitment, regulatory submissions, data collection, and safety reporting for sponsors, CROs, and academic medical centers operating at every phase of clinical research.
Get a Free Consultation
Operational Excellence
Tech Expertise
Consistent Commitment
Based on 43 Reviews
End-to-end protocol authoring and version control
Study design configuration for Phase I through Phase IV
Milestone and timeline planning with Gantt-style tracking
Amendment management and change control workflows
Budget planning and resource allocation tools
Feasibility assessment and site scoring tools
Investigator and site profile database management
Centralized site activation workflow tracking
Contract and budget negotiation management
Real-time site performance monitoring dashboards
Eligibility screening and inclusion/exclusion criteria enforcement
Digital informed consent (eConsent) management
Enrollment funnel tracking and recruitment analytics
Patient visit scheduling and adherence monitoring
Dropout and protocol deviation tracking
eTMF (electronic Trial Master File) management
IRB and ethics committee submission tracking
Regulatory authority correspondence management
FDA, EMA, and ICH-aligned document workflows
Audit-ready inspection readiness reporting
Adverse event and serious adverse event capture
MedDRA and WHO Drug Dictionary coding integration
Safety narrative generation and medical review workflows
Real-time signal detection and aggregate reporting
CIOMS and MedWatch form generation
EDC (electronic data capture) integration and management
Source data verification and remote monitoring tools
Data query management and resolution workflows
Real-time KPI dashboards for operational trial metrics
Custom statistical and regulatory reporting outputs
Support global clinical operations with intelligent automation, real-time reporting, regulatory compliance management, and cross-functional collaboration across research stakeholders.
Our enterprise-grade delivery model ensures compliance, scalability, integration readiness, and predictable execution aligned with global clinical research standards.
Assess research workflows, trial protocols, regulatory requirements, and stakeholder needs to define development scope.
Design secure and scalable architecture aligned with clinical research operations and regulatory standards.
Develop core CTMS modules and integrate with research, laboratory, and healthcare systems.
Ensure the platform meets clinical research regulations, data security standards, and operational reliability.
Deploy the platform while enabling continuous monitoring, training, and optimization for research teams.
Partner with a clinical trial management software development company that understands the science, the regulation, and the operational complexity behind every study you run.
Talk to Our Experts
Bevel is a health intelligence platform that unifies wearable and lifestyle data to generate personalized metabolic and wellness insights.
More Info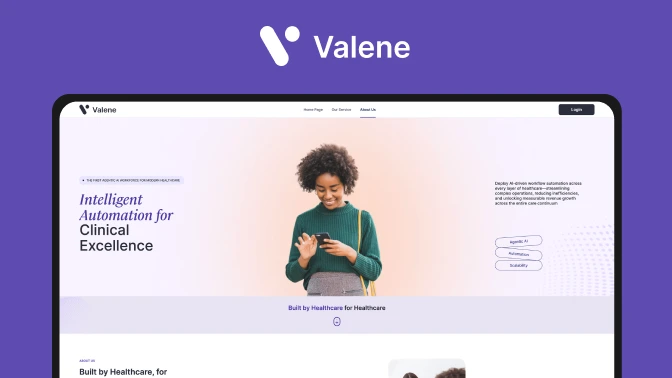
Valene Health is a pioneering telepsychiatry platform that leverages AI to enhance mental healthcare delivery.
More Info
Advinow is an AI-driven healthcare platform that automates patient engagement and consultation processes, helping healthcare providers deliver efficient, on-demand services while improving operations for urgent care.
More InfoEnterprise CTMS development typically takes 16–28 weeks depending on feature scope, integrations, regulatory requirements, and validation processes.
Yes, we design systems aligned with 21 CFR Part 11, GDPR, HIPAA, and other international regulatory frameworks.
Our solutions support seamless integration with EDC, ePRO, ERP, financial systems, and third-party clinical platforms via secure APIs.
We implement encrypted storage, audit trails, role-based access controls, and compliance-ready validation frameworks to ensure data integrity and security.
Organizations gain faster study execution, reduced administrative burden, improved compliance oversight, and enhanced cross-site operational visibility.